what the crap is ortho effect..?????plz tell me a bit!
arrange in decreasing order of acidity-----benzoic acid,o-methylbenzoic acid,p-methyl benzoic acid,m-methylbenzoic acid
-
UP 0 DOWN 0 0 19

19 Answers
The reason is , CH3 is a bulky group . When it is in the ortho positions , there is steric hindrance .
the COOH grp slightly goes out of plane , and thus weakening te delocalisation , and thus resonance stablilization of the acid is affected .
Hence it ultimately becomes more acidic .
Hope SOMEONE GET IT NOW !!
here the Active hydrogens take part in BONDING
avinav , still , i m not able to get it , how is CH3 having -R effect ??????
guys i m extremely sory...for my expln....there was an error..wrt h bonding....in that case o>benz acid> m> p..sory once again
yeah that's what i told shakespeare how would H-bonding take the charge here......i can't grasp it!!!!
dude where is hydrogen bonding coming into the picture ?????u just cant attach " H bonding " wherever u find a Hydrogen, .......and How is CH3 having a +R effect ???? pls explain me
ORTHO> BENZOIC ACID > META > PARA REASON :CH3 HAS -R AND +I EFFECT....HENCE IT INCREASES THE STABILITY OF THE CONJUGATE BASE.....SO o- BENZOIC ACID IS MOST ACIDIC....DUE TO ORTHO EFFCT O-ISOMER COMES 2ND............IN META POSN...-R EFFECT IS NULLIFIED...AND PARA FOLLWS IT..
i dont see any hydrogen bonding in o-methyl benzoic acid , since the H atom isnt attached to an effective electronegative group or u can say that C - H bond isnt sufficiently polarized
However the order is
C6H5COOH (pKa=4.2) > m-methyl C6H5COOH (pKa=4.24) > p-methyl-C6H5COOH (pKa= 4.34)
actually introduction of alkyl groups into the benzene nucleus of the carboxylic acid has a very little effect on the acidity ,
i.e p Ka values differ only by very small values .
I feel its not a reasonable question to be asked ...it seems to be much more factual one, wats d source of this question ??
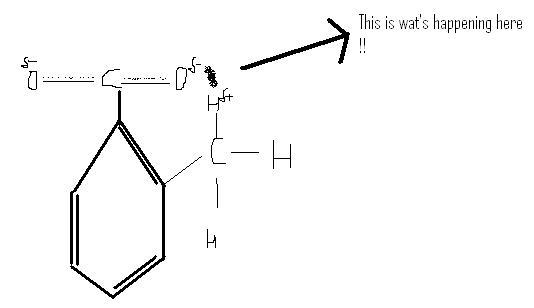
In ortho one, after making the conjugate base, there will be negative charges on oxygen's of COO-
and at ortho position there is CH3 group
so these hydrogens r hvng partial +ve charge [ hyper-conjugation]
so they will take part in intramolecular hydrogen bonding with oxygen
Making the conjugate base stable
so it will show more acidic character
this is called ortho effect.
Here the expected behavoir is :
benzoic > meta > para > ortho
BUT THE ACTUAL ORDER IS :--
ortho > benzoic > meta > para
The reason for ortho to be most acidic is the stability of it's conjugate base due to ortho effect !!
{ Or we can say INTRAMOLECULAR HYDROGEN BONDING}
then the benzoic acid
then meta and para
Para will be least stable bcoz it will destabalize the base !!
i dont have a clear idea dude...
well as far as i know, COOH group is a meta directing group.
So the effect of CH3 at ortho and para positions is the same as what an NO2 etc. group would have on the meta position of an ortho-para directed ring.
The reason why i said ortho is most acidic, is because the ortho is least stable position since COOH is meta directing.
The reason why i placed para after ortho is because it is symmetric, hence more stable than ortho.
Then i compared m-CH3-Ph-COOH and Ph-COOH. At meta position +I effect occurs, so the O-H bond of -CO-OH becomes stronger since the carbonyl group tries to abstract an electron from the ring. So the electrons bonded between C-O shift towards OH. Hence Hydrogen cannot escape as an ion easily, hence i placed Ph-COOH as more reactive than m-Met--Ph-COOH.
well here is my answer and reasoning:::::::::
decreasing order of acidity---------
benzoic acid>meta>para>ortho
reason:::at m position only +I effect of CH3 occurs,at o- and p- both accompanies +I effect[which is more pronounced at ortho position]
am i correct??????????
1. In Ph-COOH, the COOH group is meta directing.
2. CH3 is electron donating group. So it donates an electron to the Phenyl group.
3. Among ortho and para, para is more stable.
From the above observations, i think ortho is most reactive. Then follows para because it is less stable. Presence of methyl group at other positions decreases acidity i think, so COOH should follow, and finally m-methyl Ph-COOH.