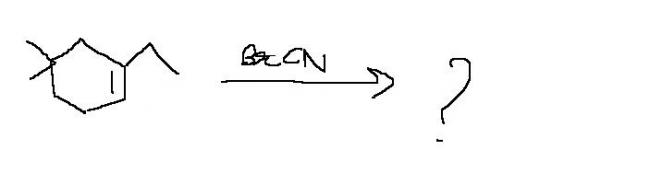
i guess ulta sankarna
cn will go on d upper carbon n br on d lower
7 Answers
°ღ•๓ÑÏ…Î
·2009-02-14 11:56:17
carbocationwill b formed teriary n cn- will go thr n br on d othr side
wild guess
°ღ•๓ÑÏ…Î
·2009-02-14 11:56:50
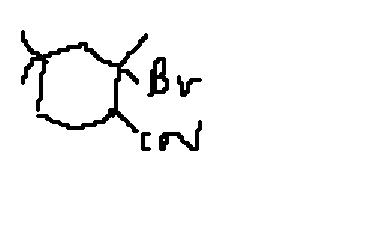
msp
·2009-02-14 12:07:20
cn molecule larger than br molecule .if cn will be at the upper carbon then it will steric hinderance which is more unstable
°ღ•๓ÑÏ…Î
·2009-02-14 12:09:48
tertairy carbocation will b forrmd coz tahts more stable
ps: sorry if ma thinkin is wrng mah org sucks
voldy
·2009-02-14 17:46:06
nice integrations is right .
sankara's product is right if he replaces Br by CN and Cn by Br.
here the reactant can be split as BrCN i.e Br+ CN-
hope it's clear