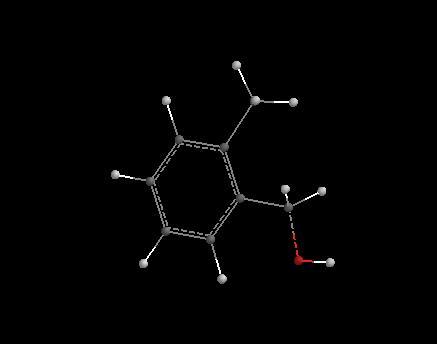
hi dude , check this out , the red mark indicates the O which attacks. the protonated acid. hope this helps. 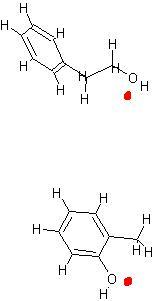
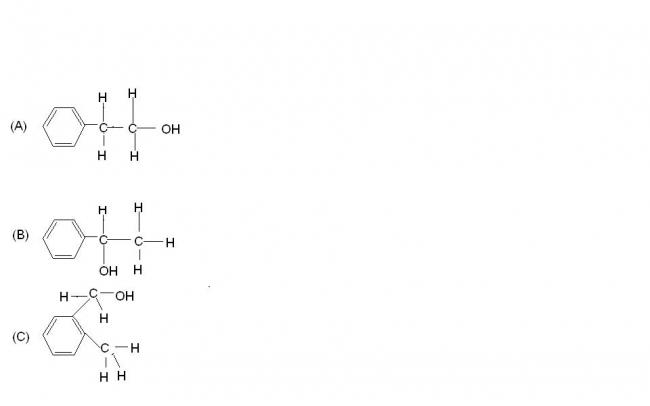 arrange the following alcohols in the increasing order of tendency towards esterification
arrange the following alcohols in the increasing order of tendency towards esterification
-
UP 0 DOWN 0 0 11

11 Answers
please give the logic behind it . I think my order is fine as in esterification , the attack of the alcohol on protonated acid is the rds , which depends on the steric factors , isn't C more sterically hinderd than A?
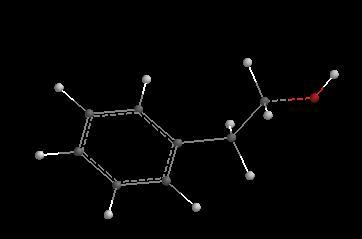
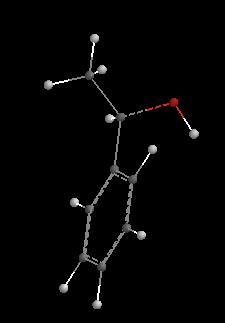
______________________________________________
______________________________________________
Now Decide....
i think it should be c>b>a (not sure)
bcoz c has most stable carbocation
yah...steric hindrance plays the role so the gp to be attached will have less free path to give the pdt..hence the order shud b
B<C<A correct me if im rong
even i think because of steric hinderance....but this is chemistry......may be there are other reasons which play an important role in deciding the order....lets think of it...