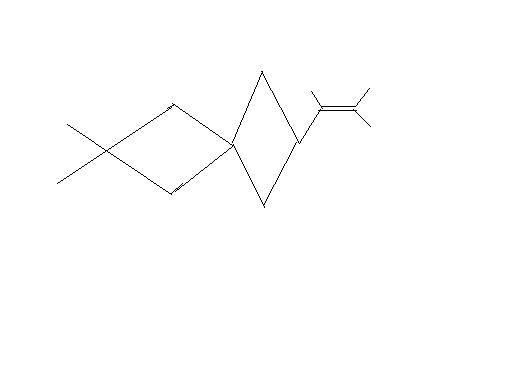
why?????????????????
ans is o dear which was also stated by subash..........
23 Answers
i know its debatable and not easily swallowable........
lets c if i get a chance i'll ask ma teacher n tell u wat he says...
u try n do da same
this is a bridge-head situation..... so i feel free rotation will b retarded....
ur point is correct that in biphenyl situation planarity is due to double bond thing.....
but ther 2 phenyls r connectged by a c-c bond......
here to v jus hav 1 carbon sufficing both the rings.......... so i m not sure!!!
i know what u r saying is similar to biphenyl situation...........but see there too there was planarity concept used due to partial double bond character due to resonance[1]
hmmmmmmmmmm[12][12].............I dont think it will be possible......maybe i am wrong...........but there is no double bond between those 2 rings....so both rings will be free to rotate....so i dont think then we can say 2 isomers..........we have to say then infinite isomers.....just like confirmation.....[2]
@Eureka :
ther r 2 geo isomers as the ther are two ring structure involved in the structuer
ther will b 2 geo isomers possible :
1. wen both the rings are coplanar!!!
2. wen both rings are perpendicular!!( as shown in the diag by subhash)
dude, i'll tell u y tom!! ( hope its fine to u)
abhi thoda isc padhna hai got che prac tom.....
c ya tom.....
no worries when eureka is here........[3]
btw nic pic bro..........[1]
how chiral??????????????????
I dont think I am sleeepy yet...........[12]
2 identical methyl groups attatched.............
yes there is no confirmational isomerism,
no cis trans becoz three identical groups about double bond
so ans will be zero.....[1]