d i think
........................................................................
.......................................................................
no the answer is not C
-
UP 0 DOWN 0 0 12

12 Answers
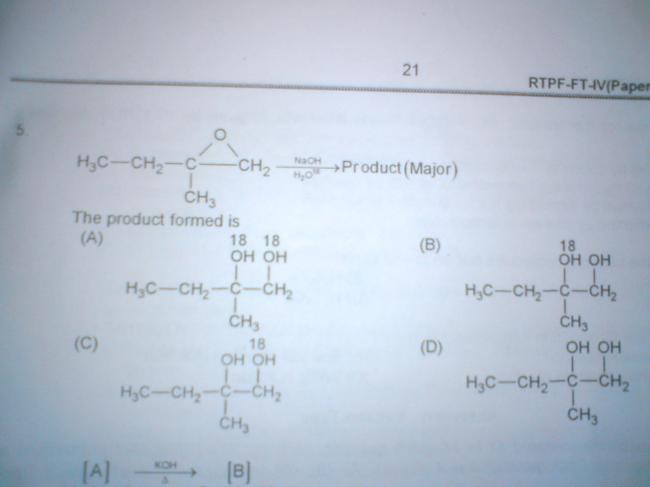
ya but why does NaOH have more preference to act as nucleophile when water is solvent and is present in larger qty?
ya but nucleophilicity of water molecule is very low it cant break epoxide ring if there would ve been a better nu as solvent then the major product would ve been changed
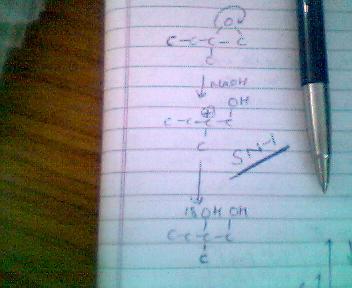
therefore the answer is B according to the SN1 mechanism in case of a 3° carbocation formation!!!!!!!!!
cheers!!!!!!!!
@aki
what you've shown takes place in acidic medium,in basic medium carbocation is formed at least sterically hindered position
d hi answer hai... to ye aki B kaise laya...
C+ banane ke baad OH- frm NaOH attack karega... na ki H2O as she had expalined in post5 ..
haan bhaiya pink kar dijiye... :P