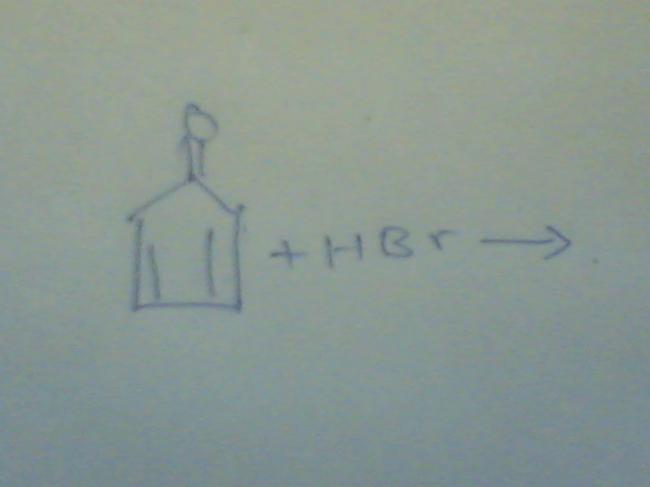i think a as aromatic character losses
14 Answers
I am not too sure but i feel C should be the ans....as in A and B an aromatic ion with a pair of pi electrons will be there...whereas in C an anti aromatic species will be formed....
sidsgr88 there is another choice in the question which also yields an anti aromatic ion but it is not the correct ans da
i think that if positive charge develops on carbon of cyclic compound in option (b) then the carbon will be become sp2 hybridised i.e. angle is 120° ,so there will be angle strain.....
just a wild guess.....
can u explain abt the option a)
can u explain with mech for ur reason.i cant get u y +ive charge develops on the carbon
i am posting the answers people please give me the mech for the products please please
thats what i was saying sankara.....in a,c,d an electrophile is added so that a carbocation is formed...then the intermediate may b classified as stable of unstable....in b two nucleophiles are removed to make it an aromatic species...