Following on from what debo said, if you go by the mechanism I think you will end up with a 5 membered ring...
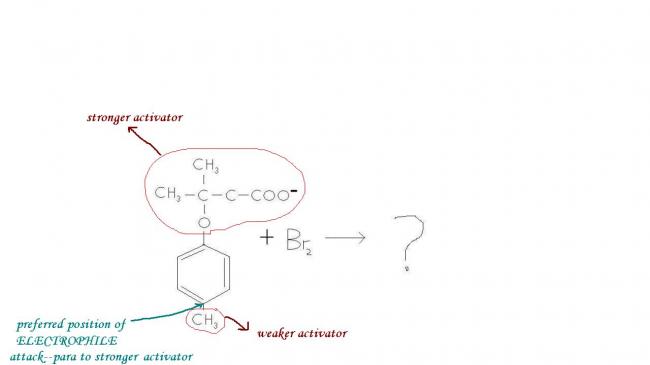
After Br gets attached, aromaticity cannot be regained as the methyl carbocation is unstable, so base cannot abstract CH3+. The positive charge is shifted by resonance to the carbon atom holding the stronger activator group, from where COO- attacks the positive charge. O- is a hard base and a carbocation is a hard acid, so they join and make merry, forming a precipitate compound so that no further resonance occurs.
6 membered ring toh ban hi nahi sakta...just resonate the positive charge and you'll see what I mean.
Okay I'm uploading an image. The possibilities are 5 and 7 membered rings, and 5 membered ring is kinetically and thermodynamically favoured.
I came across a question wich was as follows :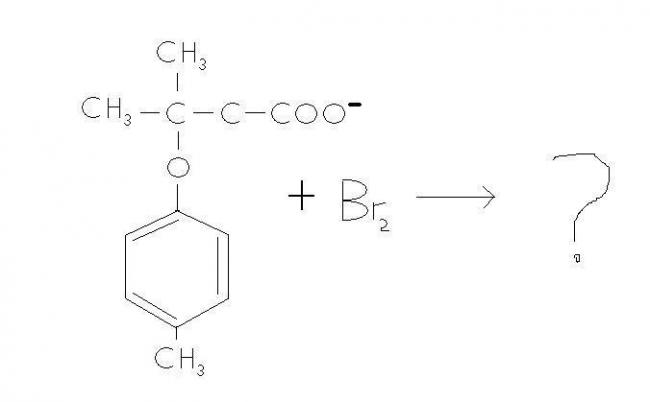
Now the product given in the ans is 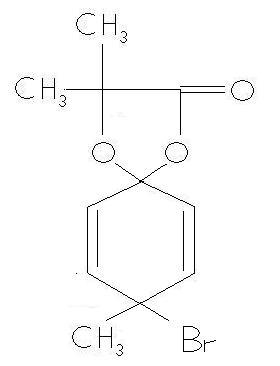
I agree that there is no problem in this answer
BUTTTTTTTTTTTTT wats wrong in the product that i m putting forward???? It has conjugation , and is a 6 membered ring !!!! where is the flaw ???? [2]
wat r ur views???
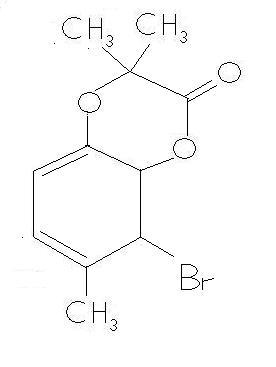
-
UP 0 DOWN 0 0 4

4 Answers
RAY
·2009-12-10 01:44:16
n i would say this is the case where the product is called abnormal product ...though it is not always abnormal....in riemer tiemenn reaction...
Pritish Chakraborty
·2009-12-17 07:23:38